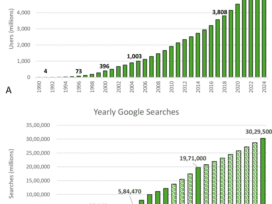
Implicity Launches SMART on FHIR App to Optimize Interoperability & Workflow Efficiencies
What You Should Know:– Implicity, a leader in remote patient monitoring (RPM) and cardiac data management solutions, released a new SMART on FHIR application to improve interoperability and workflow efficiencies for providers.– The new…
Continue Reading