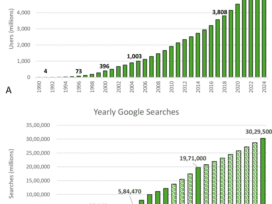
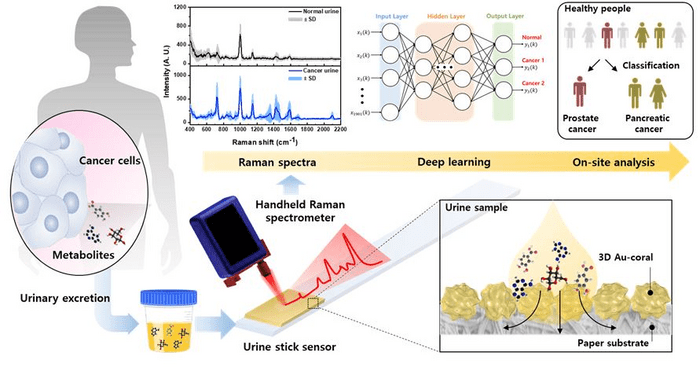
Business Intelligence in Healthcare: A Comprehensive Guide
Harness the true power of the data!
Yes. That’s the ultimate goal of Business intelligence (BI).
BI has the potential to unlock endless possibilities for the healthcare industry to thrive.
In other words, data is evolving as a new diagnostic tool in healthcare.
That’s the reason, healthcare organizations, startups, enterprises, and individuals are…