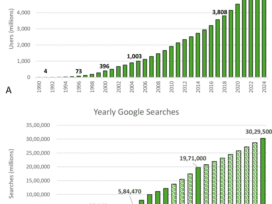
Although 2022 has brought on a greater sense of normalcy, the past few years have distracted us from one of the most overlooked epidemics plaguing healthcare: prescription drug abuse.
Before Covid-19 diverted attention away from this problem, President Trump signed on to the SUPPORT for Patients and Communities Act of 2018, instating sweeping legislation for initiatives to address the opioid epidemic. The bill, among other initiatives, requires electronic prescriptions for controlled substances (EPCS) for any covered part D drug. Although the official start of the mandate was delayed two years to January 1, 2023, the importance of this federal intervention cannot be…
Continue Reading