Roundup: Nesa gets US FDA 510(k) for fibroid mapping tool and more briefs
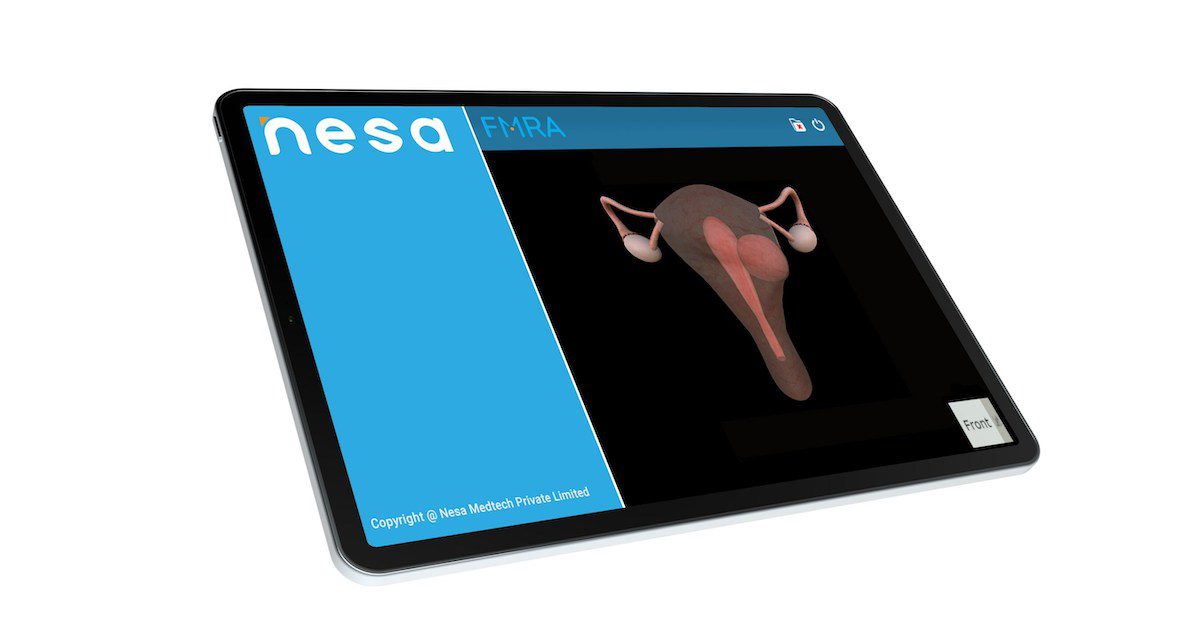
Nesa obtains US FDA 510(k) for fibroid mapping techIndian femtech Nesa Medtech has received the United States Food and Drug Administration 510(k) clearance for its fibroid mapping tool.
Its Fibroid Mapping Reviewer Application assists in…