From Reactive to Proactive: Mastering 2026 Medical Device Fleet Management
Alla K. Woodson, General Manager, Anesthesia & Global Services, GE HealthCare Patient Care Solutions
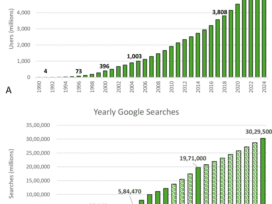
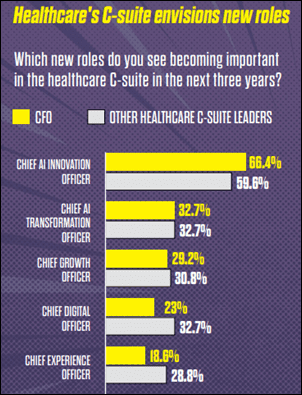
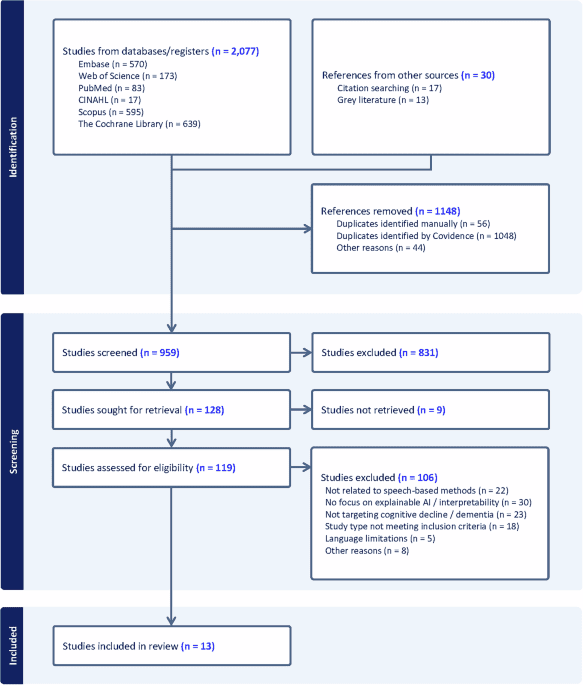
In today’s healthcare environment, hospitals typically have 10 to 15 connected medical devices per bed, meaning clinical engineering teams are often…
Continue Reading