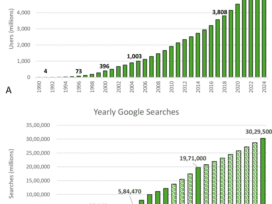
Inside eHealth 2025: Why Healthcare CIOs Should Pay Attention
What happens when patients, startups, and policymakers all show up in the same room? You get real conversations that matter to health IT leaders—and a clearer view of where the industry is headed.In this…
Continue Reading