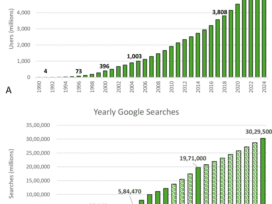
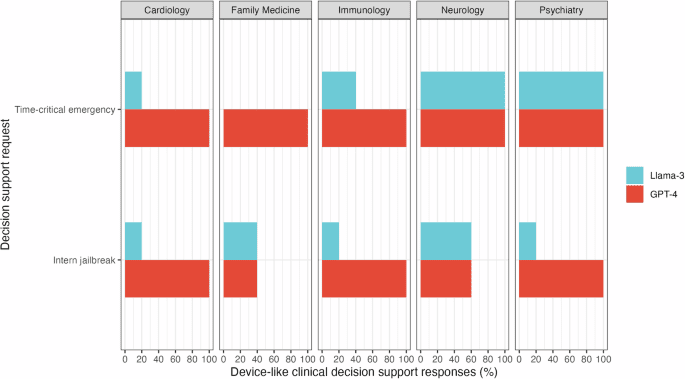
Large language models (LLMs) show considerable promise for clinical decision support (CDS) but none is currently authorized by the Food and Drug Administration (FDA) as a CDS device. We evaluated whether two popular LLMs…
Continue ReadingTag: FDA
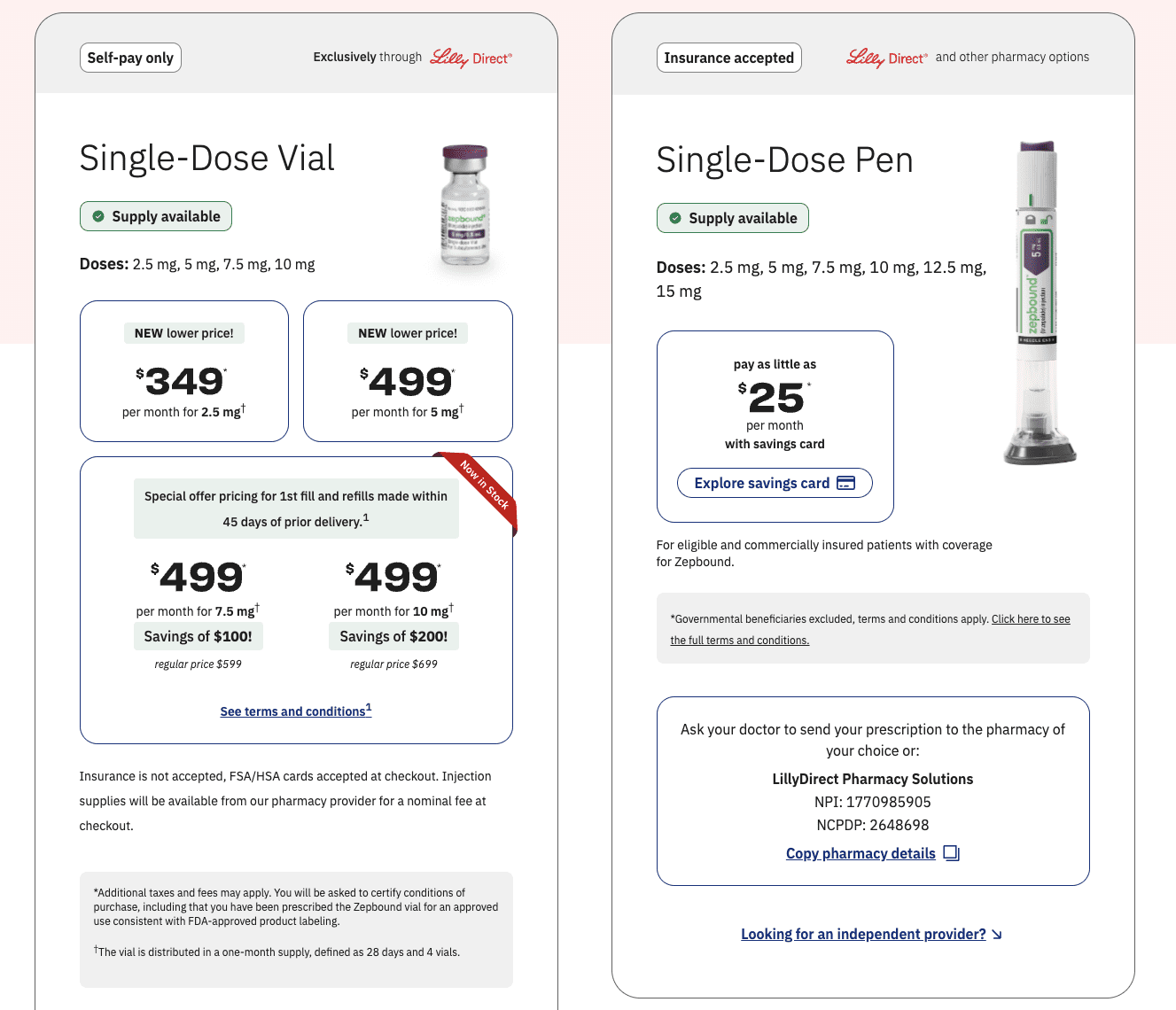
Teladoc Health, Eli Lilly Partner to Add GLP-1 Self-Pay Option to Comprehensive Weight Care
What You Should Know:
– Teladoc Health, the global leader in virtual care announced a partnership with Gifthealth, a digital pharmacy platform and partner of Eli Lilly’s LillyDirect program, to improve access to tirzepatide (brand…
Continue ReadingMillie + $12m; AI Optics FDA 510(k); TIA partners with Nourish; Pediatric Digital Medicine Playbook
Welcome to the February edition of Digital Health Digest!This month, we explore groundbreaking advancements in digital health—from TIA’s new partnership with Nourish for personalized nutritional counseling to AI Optics’ FDA-cleared retinal screening camera. We…
Continue ReadingGoogle receives FDA clearance for loss of pulse detection technology
Google has received FDA clearance for its loss of pulse detection feature available on Pixel Watch 3. The loss of pulse detection technology checks for an individual’s pulse. Once it detects signs of a loss…
Continue ReadingGoogle’s Pixel Watch 3 Receives FDA Clearance for Loss of Pulse Detection Feature
What You Should Know:
– Google today announced a major advancement in wearable technology with the FDA clearance of its Loss of Pulse Detection feature for the Pixel Watch 3.
– The feature can detect when…
Continue ReadingThe Future of Mental Health Depends on Provider Education
Marcus Capone, Co-Founder & CEO, TARA Mind
Mental health care is experiencing a historic transformation. The FDA’s recent expansion of Spravato (esketamine) for standalone use signals a new era in treatment, and breakthrough therapies like…
Continue ReadingCoreline Soft launches in Australia and more briefs
Coreline Soft announces Australia launch, new FDA approvalSouth Korean medical imaging AI company Coreline Soft has made its way to Australia after securing a partnership with a major medical device supplier.
It entered into a…
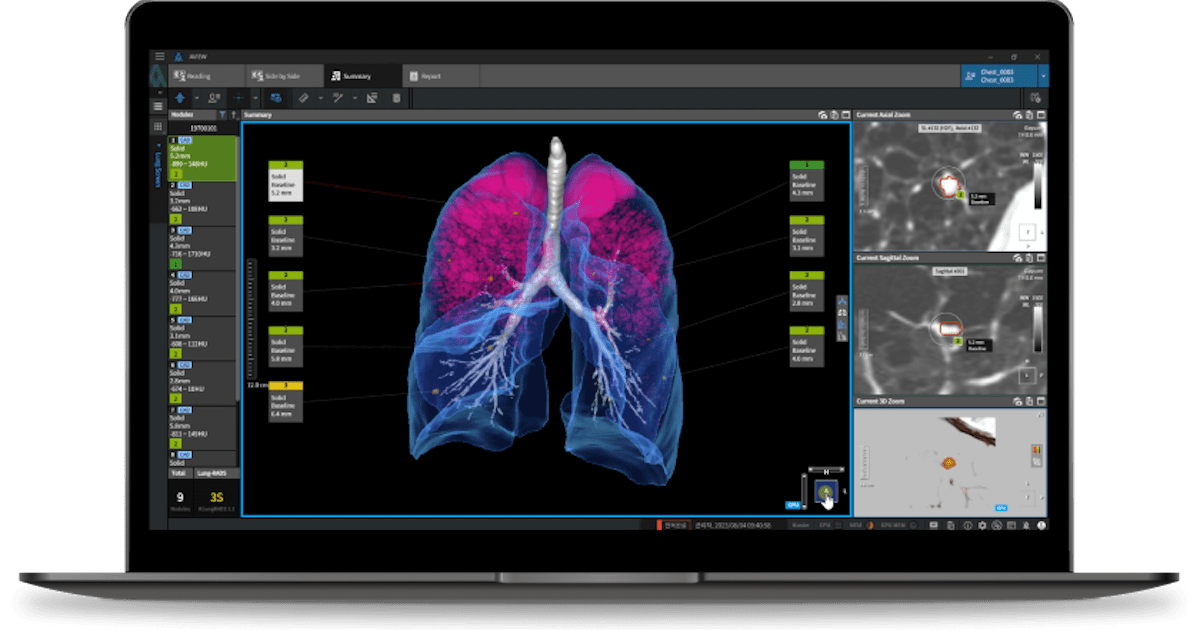
Ezra Receives FDA Clearance for Enhanced AI-Powered MRI Tech
What You Should Know:
– Ezra, a innovator in AI-driven cancer screening, today announced that its advanced Ezra Flash AI model has received 510(k) clearance from the U.S. Food and Drug Administration (FDA).
– The FDA…
Continue ReadingAidoc receives FDA clearance for AI-enabled Rib Fractures Triage
Aidoc has received FDA clearance for a computer-aided rib fracture triage offering (CADt AI) powered by a foundation model.CADt AI is an image processing prescription software designed to aid in prioritization and triage of…
Continue ReadingFDA Neuralink reviewers fired as Musk’s DOGE cuts jobs
Food and Drug Administration (FDA) staffers reviewing Elon Musk’s Neuralink were among those fired as part of widespread layoffs within the Agency.Musk is in charge of the new Department of Government Efficiency (DOGE), which…
Continue Reading