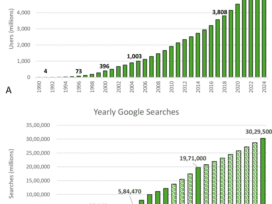
Overcoming Financial Barriers to Diversity in Clinical Trials – Q/A
Meghan Harrington, VP of Clinical Trial Financial Management at Medidata
Achieving diversity in clinical trials is crucial for developing effective and safe treatments for all populations. Yet, financial barriers often exclude underrepresented groups from participating,…
Continue Reading